Welcome to PRO Devices
Laboratory devices for measuring
INHIBITION ZONES
with automatic data registration
Developed and produced in Denmark
Automatic measurement of Inhibition Zones
PRO Devices A/S can help improve treatment of infectious diseases and thereby help save human lives by
- optimising and streamlining the manual routine workflow within Antimicrobial Susceptibility Testing (AST) using modern technology
- securing that important data are automatically saved and traceable
- helping laboratory professionals to measure all inhibitions zones and thereby meet EUCAST guidelines
Antimicrobial/Antibiotic Resistance (AMR) is growing worldwide and WHO has declared that AMR is one of the top 10 global public health threats facing humanity. Read more about AMR.
Treatment with a specific antibiotic agent can reduce the time of treatment and reduce the risk of antibiotic resistance. An AST of the infection with antibiotic disk diffusion testing and measurement of the inhibition zones gives the health staff the possibility to make an informed choice of antibiotic/s for the treatment.
Our PRO Devices software (PD-Technology) has been developed to give the health staff a tool that can raise the quality of the AST testing and make the transition to e.g. EUCAST quality standards much easier for the staff. Our products are developed in close collaboration with clinical microbiological laboratories in both Denmark and Sweden. We have been in close dialogue with people from EUCAST and from WHO during the development process.
The PD-Technology measures the size of all inhibition-zones on disc diffusion tests, at the same time the individual antibiotic discs are identified, and documentation and traceability are ensured with photo-documentation. The measuring results are automatically transferred to the LIMS system.
Please continue on the website to find out more about the benefits of our products:
- PD-Technology
- The Software capable of identifying the antibiotic discs and measuring the inhibition zones
- PD-Auto
- Under development, prototype ready for testing in a few months
- Automatic device applicable for more than 100 agar plates per day
- PD-100
- Small manual device for measuring inhibition zones
- Applicable up to 100 agar plates per day
- A good ergonomically alternative to the manual measurement
- Implemented on laboratories in Denmark and Sweden
Traceable Documentation
PD-Technology measures all inhibition zones and saves results and photos ensuring full traceability and patient safety.

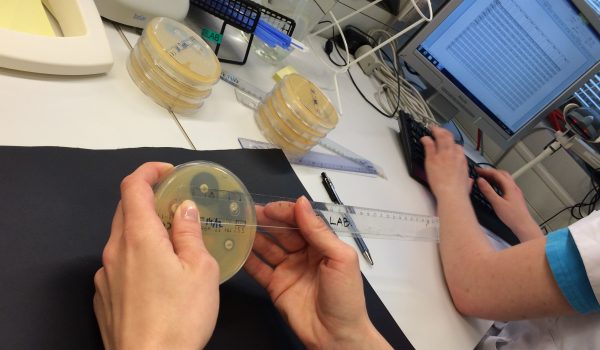
Improved ergonomics
Many laboratory professionals experience work-related problems with arms and shoulders when inhibitions zones on AST-plates are measured manually with a ruler or calliper. PD-100 and PD-Auto improves the ergonomics in the daily work.
What is Antimicrobial Susceptibility Testing (AST) and why is it important?
Antimicrobial/Antibiotic Resistance (AMR) is growing worldwide and WHO has declared that AMR is one of the top 10 global public health threats facing humanity. AMR is occurring everywhere in the world, compromising our ability to treat infectious diseases, as well as undermining many other advances in health and medicine.
Misuse and overuse of antibiotics, including the widespread habit of using broad-spectrum antibiotics, are the main drivers in the development of drug-resistant pathogens. Without effective antibiotics, the success of modern medicine in treating infections, including during major surgery and cancer chemotherapy, would be at increased risk.
The World Health Assembly in 2015 endorsed a global action plan to tackle AMR, including antibiotic resistance, the most urgent drug resistance trend.
The global action plan sets out five strategic objectives:
- to improve awareness and understanding of antimicrobial resistance;
- to strengthen knowledge through surveillance and research;
- to reduce the incidence of infection;
- to optimize the use of antimicrobial agents; and
- develop the economic case for sustainable investment that takes account of the needs of all countries, and increase investment in new medicines, diagnostic tools, vaccines, and other interventions.
A fast and effective antibiotic treatment can be crucial for patients with infections that are life threatening. Furthermore, antibiotic resistance can prolong the treatment of normally easily treated infections.

